Research
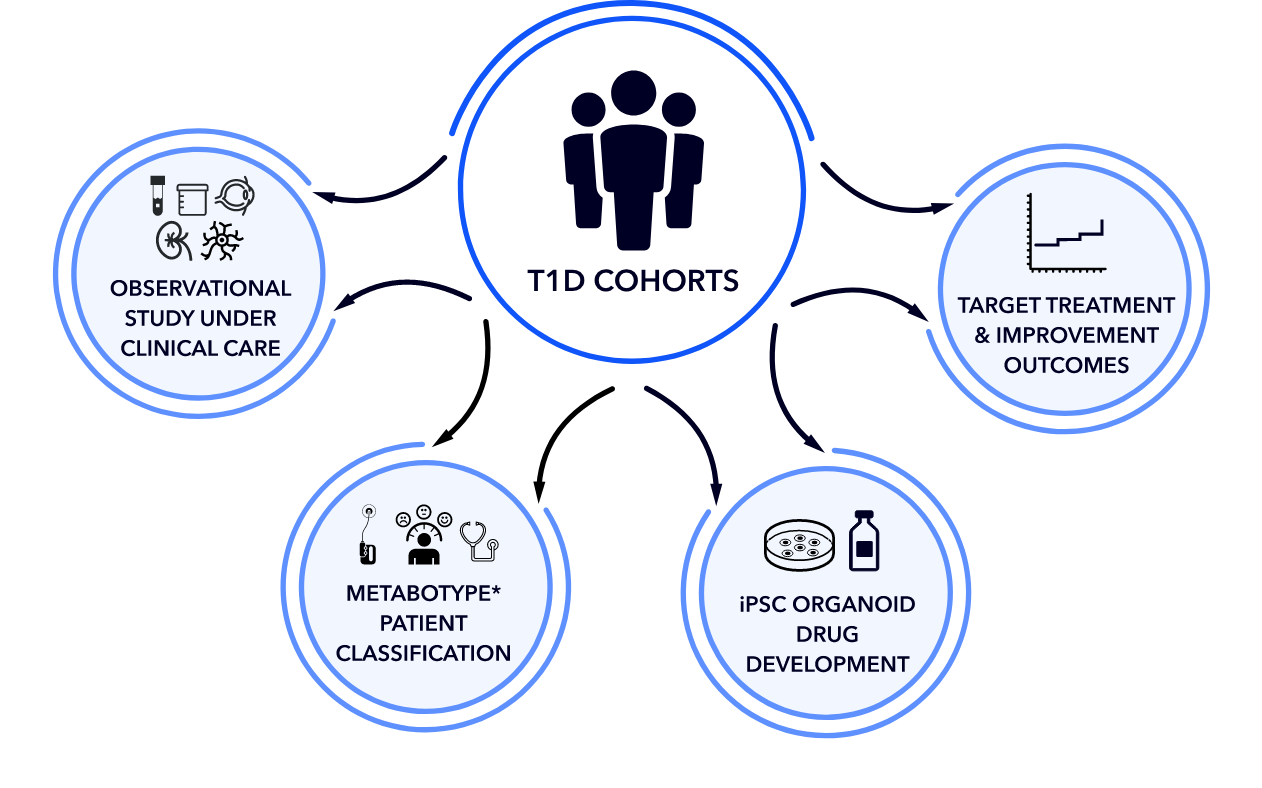
Cohorts of patients with T1D who exhibit extremes of phenotypes, such as resistance or sensitivity to long-term complications, will be analyzed for metabolic features that may explain their health. Thus, we are looking beyond HbA1c as the sole predictor of risk of reduced quality of life.
We are using this information to develop induced pluripotent organoids, models of organs that are affected by diabetes, to test for drugs that may optimize their function. This information will lead to the development of new drug treatments to improve outcomes.
Project 1:
Metabotype to optimize beta cell (ß-cell) function
Principal Investigators:
Scott Soleimanpour, MD | Lonnie Shea, PhD
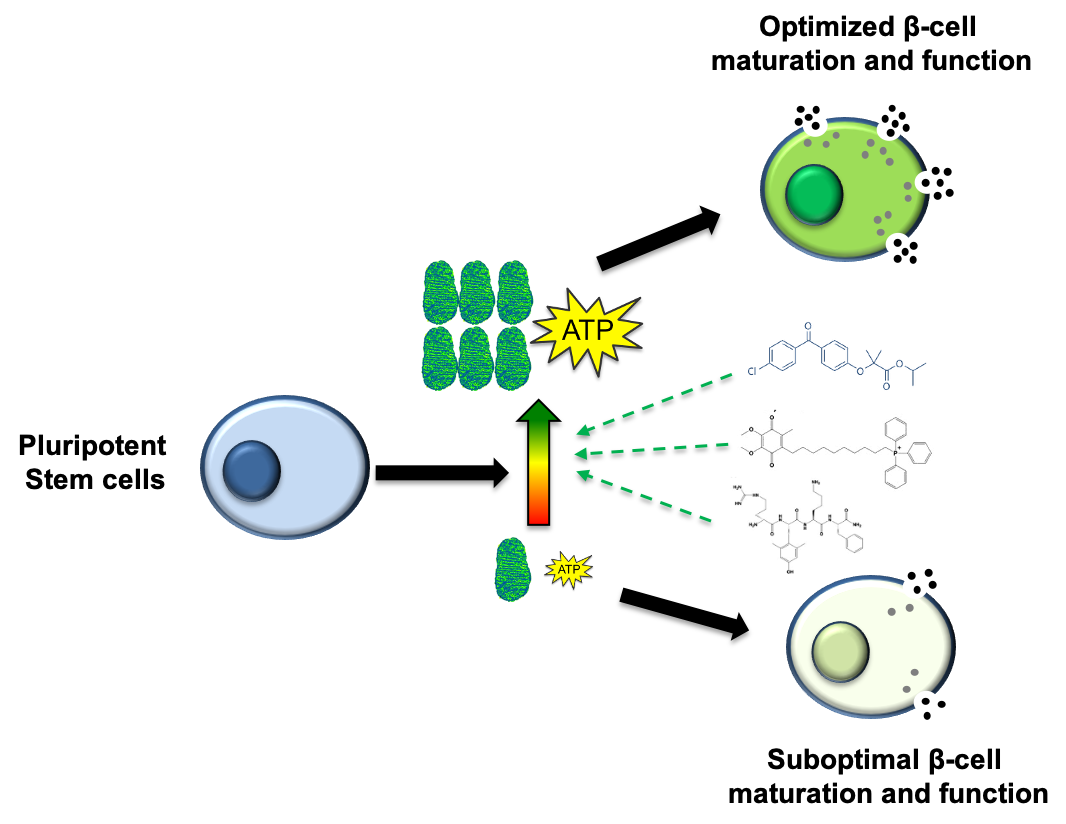
Beta cell replacement therapy is a promising approach as a cure for T1 diabetes. This type of therapy has been met with minimal success due to ineffective insulin secretion. Promising in vitro data suggests that the metabolic environment of beta cells is key to cell maturation and proper insulin secretion.
This project seeks to fully understand the beta cell maturation process with modulated metabolic environment. This complete understanding provides the foundation to improved beta cell function following transplantation into patients.
Project 2:
Employing Artificial Intelligence (AI) to reduce hypoglycemic events
Principal Investigator:
Rodica Pop-Busui, MD, PhD
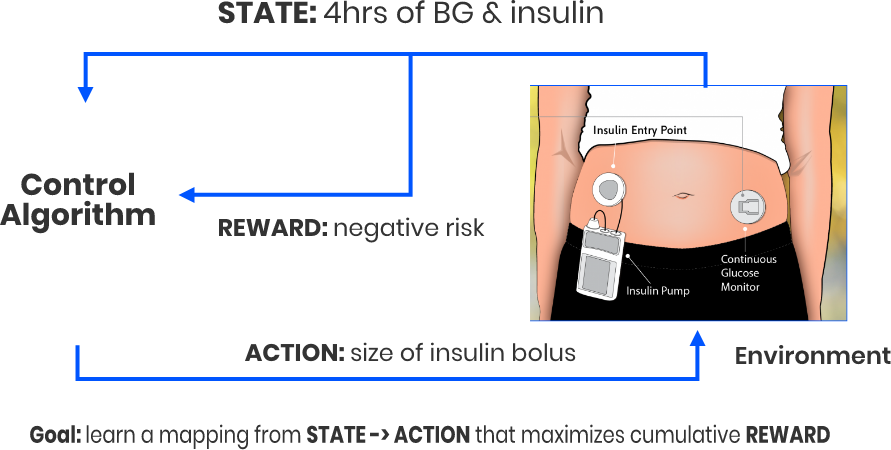
T1D patients make daily decisions based on real time blood-glucose levels with the purpose of avoiding hypoglycemic events.
We believe we can leverage the continous glucose monitoring data in conjunction with patient-specific metabolic data with reinforcement learning to create an improved AI algorithm to remove human guesswork and provide more stable blood-glucose levels.
This patient-centered project can potentially have a major impact on T1D disease burden including pyschosocial aspects of living with diabetes.
Project 3:
Identifying the metabotypes to reduce disease burden
Principal Investigators:
Subramaniam Pennathur, MD | Rodica Pop-Busui, MD, PhD
Complications and disease burden among multiple organs is a hallmark of long-term T1D. Mechanism-based therapeutic targets and predictive biomarkers having been sorely lacking for T1D patients.
This project will comprehensively define the metabolic profiles across multiple T1D cohorts for association analysis with a variety of organ complications, thus providing a data-driven path toward improved T1D patient care and reduced burden.
Potential targets and biomarkers can be tested in laboratories as the latest technologies become standards in the laboratory including testing in lab derived organs (organoids) like those derived from kidney, eye and nervous system.
Project 4:
Minimizing the burden of mental health in T1D
Principal Investigators:
Joyce Lee, MD, MPH | Rodica Pop-Busui, MD, PhD
Factors such as depression, anxiety, diabetes distress, and pain influence health outcomes and quality of life in people with type 1 diabetes. Project 4 uses two complementary approaches for looking at psychological and cognitive outcomes in patients with T1D, including a large national health care claims study, and an Ecological Momentary Assessment Study:
- To examine a large healthcare claims database to gain a deeper understanding of the utilization, costs, and burden of mental health diagnoses and their determinants for individuals with T1D.
- To examine the interplay between diabetes psychological domains (distress, anxiety, and depressive symptoms), neuropathic pain, and glycemic/metabolic outcomes using self-reported real-time psychological measures.